This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
Protein Interaction Networks
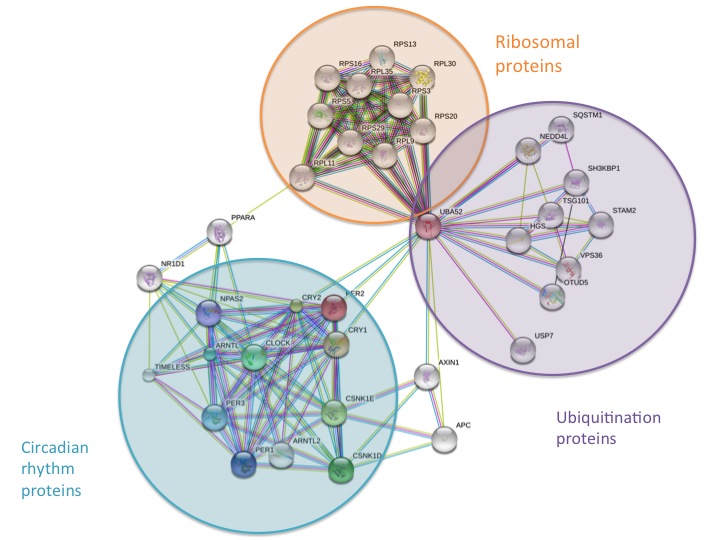
The PER2 protein has many interaction partners. Protein-protein interactions (PPIs) are crucial for biological processes [1]. They may help elucidate a protein's function or may provide insight into the genetic organization of cellular processes, information which is essential to understanding systems biology [1]. STRING is an online database of known and predicated protein interactions, including direct (physical) and indirect (functional) interactions [2]. Human PER2 results from STRING are somewhat complicated and can be seen below. PER2 is represented by a red sphere.
PER2 Interactions--Zoomed Out
Analysis
This PPI map may seem complicated at first, but PER2 interacts with three main groups of proteins at this network's level. The first group is unsurprising, a group of circadian rhythm proteins. The PER2 protein regulates itself via indirect negative regulation (it cannot directly bind to DNA). It has two transcriptional activators, Clock and Bmal1 (Clock is represented by a green sphere in the network above). Clock and Bmal1 form a heterodimer, Clock-Bmal1. Clock-Bmal1 binds to the PER2 promoter, initiating expression. PER2 enters the nucleus and disrupts the actions of Clock-Bmal1, though its exact mechanism is unknown [3]. PER2 interacts indirectly with other various circadian rhythm proteins (in the teal circle) to regulate the circadian rhythm. The second group of proteins that PER2 interacts with are ubiquitination proteins (purple circle). Ubiquitination is a post-translation modification that uses an enzyme (a type of protein that acts as a catalyst) to mark a protein with a ubiquitin [4]. Ubiquitin is a small regulatory protein that controls protein recycling and destruction [5]. It is likely that PER2 is recycled via this process. The third group of proteins that PER2 interacts with are ribosomal proteins (orange cirlce). Ribosome are an organelle that function as the powerhouse of a cell. They are responsible for protein synthesis [6].
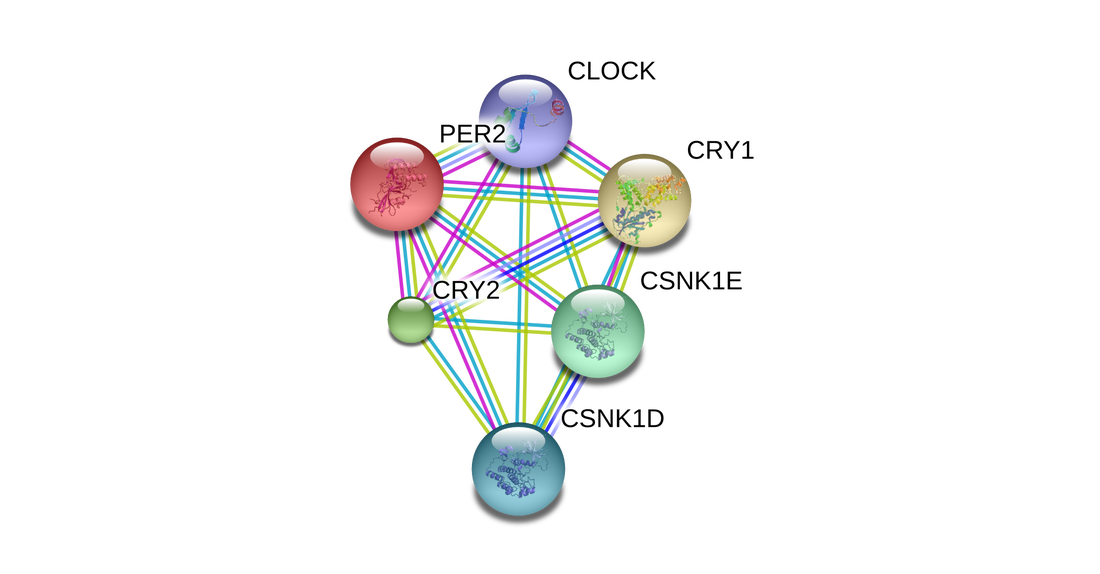
PER2 Interactions--Zoomed In
Analysis
This PPI network shows proteins that directly interact with PER2. All proteins shown in this schematic are involved with the circadian rhythm and its regulation. As mentioned above, the CLOCK protein initiates PER2 transcription by binding to its promoter. CLOCK also has histone acetyltransferase activity, which controls gene expression by chromatin remodeling. The "CRY" proteins are blue-light dependent and act together to maintain period lengths within the circadian rhythm. They may also inhibit the CLOCK protein. The CSNK1D/E proteins are casein kinase proteins who can phosphorylate many proteins and participate in the Wnt signaling pathway. They are a central component of the circadian clock and may negatively regulate its rhythm by phosphorylating PER2 [7]. It is not surprising that PER2 has such an intricate interaction network in the brain. The brain is a complex organ and PPI networks are helping tools for understanding its labyrinthine processes.
References
[1] Stelzl, U. et al. (2005). A Human Protein-Protein Interaction Network: A Resource for Annotating the Proteome. Cell. 122: 957-968. doi: 10.1016/j.cell.2005.08.029
[2] STRING http://string-db.org/
[3] Albrecht, Urs. (2007). Per2 has time on its side. Nature Chemical Biology. 3, 139-140. doi:10.1038/nchembio0307-139
[4] News-Medical.net. Ubiquitination (Ubiquitylation). http://www.news-medical.net/health/Ubiquitination-(Ubiquitylation).aspx
[5] News-Medical.net. Ubiquitin-What is Ubiquitin? http://www.news-medical.net/health/Ubiquitin-What-is-Ubiquitin.aspx
[6] Rader's Biology4Kids.com. Ribosomes-Protein Construction Teams. http://www.biology4kids.com/files/cell_ribos.html
[7] STRING. PER2 protein (Homo sapiens). http://string905.embl.de/newstring_cgi/show_network_section.pl?identifier=978060&additional_network_nodes=0&chemicalmode=-1&input_query_species=9606&interactive=no&internal_call=1&limit=5&minprotchem=0&network_flavor=evidence&previous_network_size=11&required_score=400&sessionId=8VOry0f_QP9A&targetmode=proteins&userId=bgbD8XhctF01
[1] Stelzl, U. et al. (2005). A Human Protein-Protein Interaction Network: A Resource for Annotating the Proteome. Cell. 122: 957-968. doi: 10.1016/j.cell.2005.08.029
[2] STRING http://string-db.org/
[3] Albrecht, Urs. (2007). Per2 has time on its side. Nature Chemical Biology. 3, 139-140. doi:10.1038/nchembio0307-139
[4] News-Medical.net. Ubiquitination (Ubiquitylation). http://www.news-medical.net/health/Ubiquitination-(Ubiquitylation).aspx
[5] News-Medical.net. Ubiquitin-What is Ubiquitin? http://www.news-medical.net/health/Ubiquitin-What-is-Ubiquitin.aspx
[6] Rader's Biology4Kids.com. Ribosomes-Protein Construction Teams. http://www.biology4kids.com/files/cell_ribos.html
[7] STRING. PER2 protein (Homo sapiens). http://string905.embl.de/newstring_cgi/show_network_section.pl?identifier=978060&additional_network_nodes=0&chemicalmode=-1&input_query_species=9606&interactive=no&internal_call=1&limit=5&minprotchem=0&network_flavor=evidence&previous_network_size=11&required_score=400&sessionId=8VOry0f_QP9A&targetmode=proteins&userId=bgbD8XhctF01